 An electron transport chain composed of a series of four membrane-bound protein complexes ( complexes I–IV) that catalyze redox reactions to power ATP synthesis.Oxidative phosphorylation and the electron transport chain Overview of oxidative phosphorylation and the electron transport chain Protein degradation → transamination of pyruvate to alanine by ALT in muscle cells → alanine transported to the liver → alanine transaminated to pyruvate by ALT → gluconeogenesis converts pyruvate to glucose.Glucose -alanine cycle : proteins need to be converted to glucose in the liver via gluconeogenesis.During states of catabolism (e.g., fasting, cachexia).Immediate resynthesis of ATP from ADP (very limited supply).In skeletal muscle, brain, RBCs, WBCs, kidney medulla, lens, testes, and cornea.Lactate is transported to the liver → LDH converts lactate into pyruvate → pyruvate is converted into glucose → glucose is transported to muscle cells.Lactate cannot be metabolized by muscle cells.Anaerobic glycolysis : glucose → pyruvate → lactic acid + 2 ATP.Requires O 2 and produces CO 2 and water.TCA cycle, and oxidative phosphorylation via the electron transport chain.Resting and nonstrenuous exercise states (e.g., walking).The mechanism by which ATP is produced depends on the type of activity (i.e., the energy demand) and the oxygen supply.Storage of ATP is very limited and requires constant reproduction.Main energy source when glucose is not readily available (e.g., fasting, diabetic ketoacidosis).Constantly produced in small amounts by the liver.Stored as intracellular triglycerides within the muscle cells and adipose tissue.A nonprotein calorie:nitrogen ratio of 150:1 prevents protein catabolism for energy production.Daily protein requirements: 0.8–1.0 g/kg/day.Only used during states of catabolism (e.g., fasting, cachexia).Important for muscle mass, wound healing, and intravascular oncotic pressure maintenance.Need to be converted to glucose in the liver via gluconeogenesis.Stored as intracellular glycogen (released via glycogenolysis).Sources of ATP synthesis and their caloric value In states of prolonged hypoxia (e.g., cardiac ischemia), the electron transport chain will stop running, ATP will no longer be produced, and cells may die. Oligomycin inhibits ATP synthesis by blocking the reflux of protons through ATP- synthase. Uncoupling agents, such as aspirin and 2,4-dinitrophenol, dissociate the electron transport chain from ATP synthesis by reducing the electrochemical gradient across the mitochondrial membrane. 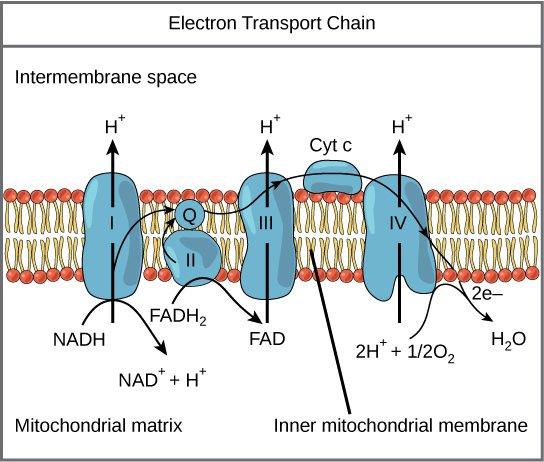
Re-entry of these protons through ATP- synthase ( complex V) into the mitochondrial matrix results in the phosphorylation of adenosine diphosphate ( ADP) into ATP. The electron carrier complexes not only transfer electrons, but also pump protons out of the mitochondrial matrix into the mitochondrial intermembrane space, thereby creating an electrochemical gradient. The electron transport chain ( mitochondrial respiratory chain) is embedded in the inner mitochondrial membrane and consists of four electron carrier complexes ( complexes I–IV) that transfer electrons from nicotinamide adenine dinucleotide ( NADH) and flavin adenine dinucleotide ( FADH 2) to oxygen, thereby generating water (H 2O). The process takes place within the mitochondria and involves oxidation-reduction reactions and the generation of an electrochemical gradient by the electron transport chain. Oxidative phosphorylation is a metabolic pathway through which cells release the energy stored in carbohydrates, fats, and proteins to produce adenosine triphosphate ( ATP), the main source of energy for intracellular reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
